ISASS Policy Guideline– Surgical Treatment of Lumbar Disc Herniation with…
- Home
- Public Policy
- ISASS Policy Guideline– Surgical Treatment of Lumbar Disc Herniation with Radiculopathy
- Categories
- Tags
ISASS Policy Guideline – Surgical Treatment of Lumbar Disc Herniation with Radiculopathy
Morgan Lorio, MD, FACS,1 Choll Kim, MD, PhD,2 Ali Araghi, DO,3 Jason Inzana, PhD,4
James J. Yue, MD51 Advanced Orthopedics – Altamonte Springs, Florida, 2 Spine Institute of San Diego – San Diego, California, 3 The CORE Institute – Sun City West, Arizona, 4 Telos Partners, LLC – Denver, Colorado, 5 CT Orthopaedics; Frank H. Netter School of Medicine – Hamden, Connecticut
Abstract
Lumbar disc herniation is a frequent cause of low back pain and radiculopathy, disability, and diminution in quality of life. While nonsurgical care remains the mainstay of initial treatment, symptoms that persist for prolonged periods of time are well-treated with discectomy surgery. A large body of evidence shows that, in patients with unremitting symptoms despite a reasonable period of nonsurgical treatment, discectomy surgery is safe and efficacious. In patients with symptoms lasting greater than 6 weeks, various forms of discectomy (open, microtubular, and endoscopic) are superior to continued nonsurgical treatment. The small but significant proportion of patients with recurrent disc herniation experience less improvement overall than patients who do not experience reherniation after primary discectomy. Lumbar discectomy patients with large annular defects (≥6 mm wide) are at a higher risk for recurrent herniation and revision surgery. Annular closure via a bone-anchored device has been shown to decrease the rate of recurrent disc herniation and associated reoperation in these high-risk patients. After a detailed review of the literature, current clinical evidence supports discectomy (open, microtubular, or endoscopic discectomy) as a medically necessary procedure for the treatment of LDH with radiculopathy in indicated patients. Furthermore, there is new scientific evidence that supports the use of bone-anchored annular closure in patients with large annular defects, who are at greater risk for recurrent disc herniation.
Introduction
Lumbar disc herniation (LDH) is a localized displacement of intervertebral disc material beyond the normal perimeter of the disc space.1,2 Depending on the volume, location, and type of herniation, adjacent neural structures may become compressed, resulting in radiculopathy associated with pain, numbness, or weakness.3,4 Impingement of the lumbosacral nerves by a herniated disc is the leading cause of sciatica, which is characterized by low back pain and radiating leg pain.5
Patient history and physical examination are the most important steps in the diagnosis of LDH with radiculopathy.1 The physical examination may include manual muscle testing, sensory testing, supine straight leg raise, Lasegue’s sign and crossed Lasegue’s sign.1 Other functional tests (e.g. Bell test, hyperextension test, reflex tests, nerve stretch test, and lumbar range of motion test) have varying degrees of sensitivity and specificity, but can be incorporated depending on a physician’s experience and preference.1,6 If the patient history and physical examination results are consistent with symptomatic LDH, noninvasive visualization of the neural structures is used to confirm disc herniation. Magnetic resonance imaging (MRI) is considered the most appropriate modality to confirm LDH, but computed tomography (CT) with or without myelography should be considered if MRI is contraindicated or inconclusive.7,8
Many patients with symptomatic LDH, particularly those with mild or moderate symptoms, will improve with nonsurgical management, which may include manual spinal manipulation, physical therapy, nonsteroidal anti-inflammatory drugs, analgesics, and/or corticosteroid injections.1,9,10 Results from randomized controlled trials (RCT) have shown that 35-54% of patients assigned to continued nonsurgical care undergo discectomy within 6 months due to persistent or aggravating symptoms.11-14 A survey of 814 neurosurgeons found that 46% of respondents recommended a minimum of 4 to 8 weeks of nonsurgical management before surgery is indicated; 34% recommended 8 weeks or more and 20% recommended less than 4 weeks.15 Based on a health technology assessment, which included a systematic literature review, public feedback, and voting from a clinical committee, the Washington State Health Care Authority requires at least six weeks of nonsurgical care unless progressive motor weakness is present.16,17 Thus, the decision to continue nonsurgical care or undergo surgery considers the patient history, physical examination, radiological findings, and severity of symptoms.
When surgery is indicated, discectomy is the primary surgical intervention for LDH with radiculopathy.18 The principal goals of discectomy are to surgically remove the displaced disc material, decompress the affected neural structures to relieve symptoms, and minimize the chance of recurrence by removing loose or degenerated material from the intervertebral space and surrounding area. Discectomy can be performed through an open, mini-open (e.g. microtubular), or endoscopic approach. Each of these methods enables direct, light-based visualization, in contrast to percutaneous techniques that solely utilize intra-operative radiological imaging for indirect visualization.
Upon completion of the discectomy, prior to closure, there are currently no widely accepted practices for repair of the annulus fibrosus. Studies have shown that large (≥ 6 mm wide), unrepaired annular defects increase the risk of symptom recurrence and additional surgery.19,20 These data have led to the development of several annular closure or repair devices to help reduce the risk of recurrence and reoperation in this high-risk patient population. Most of those techniques and closure systems lacked clinical evidence and are no longer commercially available. A bone-anchored implant is FDA-approved and has clinical evidence demonstrating effectiveness for large annular defects.
The aim of this ISASS Policy Statement is to discuss the evidence from RCTs, prospective cohort studies, and recent meta-analyses to inform treatment and coverage recommendations for primary discectomy and annular repair in indicated patients with LDH and radiculopathy.
Summary of the Clinical Evidence
Discectomy versus nonsurgical care for LDH with radiculopathy
Several RCTs and prospective cohort studies have shown that discectomy provides faster and/or greater pain relief, recovery from disability, and patient satisfaction compared to continued nonsurgical care (Table 1).11-14,21,22 Clinical evidence also indicates that discectomy-related surgical complications are rare. The most common complications include dural tears (1% to 4% of cases),11,13,14,21,23 blood transfusions (2% to 3% of cases),14,21,23 and neural injuries (0.4% of cases).21 None of the RCT or prospective cohort studies reported cases of perioperative mortality,11-14,17,21-25 indicating that the risk of discectomy-related death is negligible. A recent (2019) analysis of 34,639 LDH operations performed over 15 years found that complications occurred in only 2.7% (95% confidence interval: 2.6% to 2.9%) of cases, with no perioperative deaths.18
Table 1. Summary of clinical studies comparing discectomy vs. nonsurgical care
Study Citation
Patient Population Study Design
Sample Size
Follow-up Time
Crossover Rate
Results
NS to S S to NS Atlas et al 1996 21 Sciatica + failed ≥ 2 wks of NS care; mean ages were 42 (18-85) (S) and 42 (19-84) (NS) yrs Prospective Cohorts
NS: 232 S: 275
1 y %* 0% Increased proportion of S patients reported improved back pain (75% vs 54.5%; P<0.001), leg pain (81.3% vs 55.8%; P<0.001), change in predominant symptom (30.6% vs 11.4%; P<0.001), and satisfaction (61.1% vs 46.0%; P<0.005) compared to NS patients; of all S patients, 3.4% had >500cc intraoperative bleeding, 2.2% had dural tear, and 0.4% had neural injury Atlas et al 2001 26 5 yrs N/A N/A Compared to NS patients, S patients had greater improvements from baseline in back pain (-2.2 vs -1.0; P<0.001), leg pain (-3.7 vs -1.7; P<0.001), and sciatica (-11.7 vs -4.6; P<0.001); predominant pain symptom improved significantly more in S patients (70% vs 56%; P=0.005); satisfaction with treatment was greater in S group (63% vs 43%; P=0.005) Atlas et al 2005 27 10 yrs N/A N/A Patients treated with S had greater improvements from baseline in leg pain (-3.5 vs -0.3; P=0.02), symptom index (-9.6 vs -0.2; P=0.02), and disability (-7.3 vs -1.2; P=0.02; Roland scale) Butterman 2004 13 Symptomatic + LDH that encompassed >25% of spinal canal + failed ≥ 6 wks of NS care; mean ages 40 (S) and 41 (NS) yrs RCT NS: 50 S: 50
3 yrs 54% 0% Compared to NS patients, S patients had significantly improved leg pain, bodily pain, disability, and motor deficit scores at 1 to 3 months; leg pain significantly improved with S at 4 to 6 months; of all S patients, 3% had incidental durotomies, 3% had spinal arthrodesis, and 1% had a seroma Osterman et al 2006 12 6 to 12 wks of radicular pain + CT scan + physical exam; mean ages 37 (S) and 38 (NS) (20-50 for all) yrs RCT NS: 28 S: 28
2 yrs 39% 0% ITT analysis indicated that the S group had reduced leg pain scores (12 vs 25; P<0.01) at 6 wks follow-up; satisfaction with treatment was increased with S through 2 yrs; 1 patient in the S group had urosepsis that required antibiotics Weinstein et al 2006 14 # Radicular pain + SLR test or neurologic deficit + failed ≥ 6 wks of NS care; mean ages 42 ± 12 (S) and 43 ± 11 (NS) yrs RCT NS: 256 S: 245
2 yrs 45%
40% As-treated analysis demonstrated significantly improved body pain, physical function, disability, and sciatica with S compared to NS at all follow-up times through 2 yrs; for example, significantly improved body pain (TE: 15; 95% CI: 10.9 to 19.2), physical function (TE: 17.5; 95% CI: 13.6 to 21.5), disability (TE: -15; 95% CI: -18.3 to -11.7), and sciatica (TE: -3.2; 95% CI: -4.3 to -2.1) with S at 1 y; operative complications include dural tears (4% of cases) and blood transfusions (2% of cases) Weinstein et al 2006 23 # Confirmatory signs of lumbar radiculopathy for ≥ 6 wks; mean ages 41 ± 11 (S) and 44 ± 12 (NS) yrs Prospective Cohorts NS: 191 S: 528
2 yrs 22% 4% Significantly improved bodily pain (TE: 10.2; 95% CI: 5.9 to 14.5), physical function (TE: 12; 95% CI: 7.9-16.1), disability (TE: -13.4; 95% CI: -17 to -9.7), sciatica (TE: -2.1; 95% CI: -3.2 to -1.0), and satisfaction (TE: 11.7; 95% CI: 4.5 to 18.9) index scores with S compared to NS at 2 yrs; most common operative complication was dural tears (2% of cases); 2 S patients required transfusions Weinstein et al 2008 28 # Confirmatory signs of lumbar radiculopathy for ≥ 6 wks; mean age of combined cohorts 41.7 yrs Combined Prospective Cohort and RCT NS: 478 S: 766
4 yrs N/A N/A As-treated analysis demonstrated significantly improved leg pain (TE: -0.9; 95% CI: -1.2 to -0.6), back pain (TE: -0.8; 95% CI: -1 to -0.5), body pain (TE: 15; 95% CI: 11.8 to 18.1), physical function (TE: 14.9; 95% CI: 12 to 17.8), disability (TE: -13.2; 95% CI: -15.6 to -10.9), and sciatica (TE: -3.3; -4.1 to -2.5) with S compared to NS at 4 yrs follow-up Lurie et al 2014 29 # 8 yrs N/A N/A As-treated analysis demonstrated significantly improved leg pain (TE: -0.6; 95% CI: -0.9 to -0.4), back pain (TE: -0.5; 95% CI: -0.7 to -0.2), body pain (TE: 10.9; 95% CI: 7.7 to 14), physical function (TE: 10.6; 95% CI: 7.7 to 13.5), disability (TE: -11.3; 95% CI: -13.6 to -9.1), and sciatica (TE: -1.8; -2.6 to -1) with S compared to NS at 8 yrs follow-up Peul et al 2007 11 Radiologically confirmed LDH + radiculopathy for 6 to 12 wks; mean ages 42 ± 10 (S) and 43 ± 10 (NS) yrs RCT NS: 142 S: 141
1 y 39% 11% ITT analysis reported improved leg pain (MD: 17.7; 95% CI: 12.2 to 23.1), back pain (MD: 11.3; 95% CI: 5.6 to 17.4), disability (MD: 3.1; 95% CI: 1.7 to 4.3) and recovery (MD: 0.9; 95% CI: 0.6 to 1.2) with S compared to NS at 8 wks of follow-up; no difference at 1 y; complications occurred in 1.6% of S patients: 2 dural tears and 1 wound hematoma Thomas et al 2007 25 Leg pain and/or weakness + MRI or CT scan; mean ages 42 ± 13 (S) and 44 ± 12 (NS) yrs Prospective Cohorts
NS: 164 S: 333
1 y 2% 0% ITT analysis detected no difference in NASS questionnaire for neurogenic symptoms (44.6 NS vs 43.8 S) at 2 yrs follow-up; statistically significant difference favoring S for NASS pain and disability (PDS), and the mental health components of the SF-36; no adverse events were reported McMorland et al 2010 24 Leg-dominant symptoms + evidence of nerve compression on MRI + failed ≥ 3 months of NS care; mean ages 42 (S) and 42 (NS) yrs RCT NS: 20 S: 20
1 y 40% 15% ITT analysis detected no difference in pain, disability, or SF-36 indices over 52 wks of follow-up; no adverse events were reported Gugliotta et al 2016 22 Radicular pain + nerve root irritation + MRI or CT confirmation; mean ages 50 ± 14 (S) and 50 ± 13 (NS) yrs Prospective Cohorts
NS: 73 S: 297
2 yrs N/R N/R NASS pain score was significantly reduced by S compared to NS at 6 wks (MD: -1.0; 95% CI: -1.9 to -0.1); no difference between S and NS at 12 wks (MD: -0.3; 95% CI: -1.3 to 0.6), 1 y (MD: -0.2; 95% CI: -1.2 to 0.8), and 2 yrs (MD: -0.1; 95% CI: -1.2 to 1.0) follow-up; no adverse events were reported Abbreviations: NS, nonsurgical care group; S, surgery group; RCT, randomized controlled trial; ITT, intention-to-treat; SLR, straight leg raise; CT, computerized tomography; MRI, magnetic resonance imaging; LDH, lumbar disc herniation; TE, treatment effect; CI, confidence interval; MD, mean difference between NS and S; NASS, North American Spine Society; SF-36, Short-Form (36) Health Survey; y, year; yrs, years; wks, weeks; N/A, not applicable; N/R, not reported * In this study, 14.7% (n=40) of NS care patients crossed over to S before 3 months follow-up and were included in the S group during analysis. Another 6.9% (n=19) crossed over after 3 months and were included in the NS group.
# These studies report 2-, 4-, and 8-year follow-up timepoints and outcomes from the SPORT RCT, prospective cohorts, and combined cohorts.
Early evidence on the efficacy of discectomy for symptomatic LDH came from a large, multicenter prospective cohort study called the Maine Lumbar Spine Study (MLSS).21 The MLSS involved 275 patients treated surgically and 232 treated with nonsurgical care. At the 1-year follow-up, patients treated with discectomy reported greater improvement in back and leg pain (P<0.001), functional status (P<0.001), and overall satisfaction (P=0.005) compared to nonsurgical care.21 Additionally, a larger percentage of patients in the discectomy group reported complete relief from their predominant symptoms (31% vs 11%; P<0.001). Long-term follow-up analyses in the MLSS reported that these greater improvements among discectomy-treated patients persisted over 5 years for leg pain, back pain, function, and satisfaction (P≤0.001) and over 8 to 10 years for leg pain and function (P<0.05).26,27
The Spine Patient Outcomes Research Trial (SPORT) is a large, NIH-funded, multicenter RCT and an observational cohort study comparing discectomy versus nonsurgical care in patients with lumbar radiculopathy secondary to LDH. The RCT included 501 patients with confirmed symptomatic LDH treated with discectomy (n=245) or nonsurgical care (n=256).14 Patients eligible for surgery had radicular pain, a positive straight leg test or neurologic deficit, failed at least 6 weeks of nonsurgical care, and had radiologically confirmed LDH. In the RCT arm of the study, 45% of patients randomized to nonsurgical care crossed over to surgery and 40% of patients randomized to surgery chose to continue with nonsurgical care. Furthermore, patients that crossed over to surgery had significantly worse baseline scores for Short Form 36 (SF-36) bodily pain (P=0.03) and function (P<0.001), Oswestry Disability Index (ODI; P<0.001), sciatica frequency (P=0.009), and sciatica bothersomeness (P=0.001). These confounding events likely explain why the intent-to-treat (ITT) analysis did not detect significant differences in pain, function, or disability between treatment groups. ITT analysis is intended to reduce the risk of bias by evaluating results according to the randomized treatment allocation, irrespective of the treatment that patients actually received.30 In RCTs with high crossover or non-adherence rates, ITT analyses are difficult to interpret and an as-treated analysis is recommended to determine the treatment effect.31 In the SPORT RCT, an as-treated analysis evaluated outcomes based on the adjusted covariates of time and treatments actually received.14 Those results showed significantly better improvements in the SF-36 pain and function scores, ODI, sciatica scores, as well as higher patient satisfaction, at all timepoints in the discectomy group compared to nonsurgical care.14
In addition to randomized patients, SPORT also included observational cohorts comprised of patients that declined to be in the RCT.23 In the observational cohorts, 528 patients were treated with discectomy and 191 patients received nonsurgical care over the first 2 years. Compared to the nonsurgical group, patients in the discectomy group had significantly greater improvements in primary outcomes (i.e. bodily pain, physical function, and ODI) and secondary outcomes (i.e. sciatica bothersomeness, satisfaction, and self-rated improvement) over 2 years of follow-up (P<0.001 for all metrics).23 An as-treated subgroup analysis of combined observational and randomized cohorts demonstrated that all 37 subgroups experienced significantly greater improvements with discectomy compared to nonsurgical care (P<0.05).32 Some of the subgroups with the greatest treatment effect from discectomy included older patients (age > 41 years), those with deteriorating symptoms at baseline, and those without joint problems.32 Longer term follow-up studies of the combined SPORT cohorts confirmed that the benefits of discectomy persisted through 8 years.28,29,33 As-treated analyses at 8 years follow-up adjusted for potential covariates, such as demographics, smoking status and LDH location, and demonstrated significant benefits in favor of discectomy for all primary and secondary outcomes of pain, function, sciatica bothersomeness, patient satisfaction, and self-rated improvement (P<0.001 for all metrics).33 Together, the SPORT study as a whole represents robust and high quality clinical evidence in support of discectomy compared with nonsurgical care in both short- and long-term benefits for treatment of LDH with radiculopathy among indicated patients.
Three more RCTs and 1 prospective cohort study demonstrated positive outcomes with discectomy compared to nonsurgical care at early time points.11-13,22 Peul et al. conducted a multicenter RCT comparing 141 patients treated with discectomy versus 142 patients treated with continued nonsurgical care.11 All patients had radiculopathy and radiologically confirmed LDH. Thirty-nine percent of patients assigned to nonsurgical care crossed over to discectomy within 1 year and 11% assigned to surgery continued with nonsurgical care. Based on ITT analysis, discectomy resulted in greater symptom relief at early timepoints of 2 to 12 weeks. By 1 year, the differences were not statistically significant; however, this finding, based on ITT analysis, is confounded by the high crossover rate from nonsurgical care to discectomy. The authors did not report analysis of the as-treated populations.
Butterman conducted a single-site RCT comparing 50 patients treated with discectomy versus 50 patients treated with epidural steroid injections.13 All patients had clinical symptoms of LDH, radiologically confirmed disc herniation that encompassed more than 25% of the spinal canal cross-sectional area, and failed at least 6 weeks of nonsurgical care. Twenty-seven (54%) patients failed epidural steroid injection treatment and crossed over to discectomy. The results demonstrated that discectomy significantly improved leg pain, bodily pain, ODI, and neurological deficit scores 1 to 3 months after surgery (P<0.05), suggesting an early clinical benefit of discectomy. After 3 months, outcomes measures were not statistically different between treatment groups; the only exception was that leg pain was improved with discectomy at 4 to 6 months (P<0.05).
Österman et al. conducted a small RCT analyzing 56 patients: 28 treated with discectomy and 28 with nonsurgical care.12 Similar to SPORT and Peul et al., 39% of patients randomized to nonsurgical care underwent surgery by 1 year. All patients allocated to the surgery group underwent discectomy. Results from the ITT analysis showed that patients treated with discectomy had significantly greater patient satisfaction through 2 years of follow-up and an early advantage in the reduction of leg pain at 6 weeks follow-up. Additional trends in favor of discectomy were noted; however, interpretation is limited by the small sample size and large number of crossover patients.
Recently, in a prospective cohort study, Gugliotta et al. reported that patients treated with discectomy had significantly reduced North American Spine Society (NASS) questionnaire back pain at 6 weeks compared to nonsurgical treatment, indicating a faster improvement in pain with discectomy.22 No differences were observed at later time points; however, 42% of patients in the surgery group and 55% in the nonsurgical care group were lost to follow-up, which limits the data quality at later time points.
In contrast, one RCT of 40 patients and one prospective cohort study of 497 patients concluded that there were no significant differences in patient outcomes between discectomy and nonsurgical care for symptomatic LDH at 1 year.24,25 Interpretation of the RCT was limited by a small sample size, large rate of cross-over (40%) in the ITT analysis, and a lack of as-treated analysis.24 In the cohort study, patients treated with discectomy and nonsurgical care experienced a similar, nonsignificant (P=0.58) improvement in the primary outcome (i.e. NASS neurogenic symptoms score) after adjusting for covariates.25 However, the discectomy group achieved significantly greater improvements in the pain and disability scores as well as the mental component summary (MCS) of the SF-36 (P<0.05), even after adjustment for covariates.25
A 2019 systematic review and meta-analysis examined 11 studies (3,232 patients) comparing discectomy versus nonsurgical care.34 Discectomy was found to be more effective than nonsurgical care in significantly reducing leg pain (P<0.001) and back pain (P=0.02). The authors also reported a trend towards greater reductions in disability after discectomy, but the comparison was not statistically significant (P=0.09). Comparison of any surgical intervention with nonsurgical care can be challenging and such studies require careful interpretation (e.g. ITT vs. as-treated analyses with adjustment for covariates). While studies of discectomy versus nonsurgical care can be confounded by high crossover rates and expectation bias,35 the as-treated analyses consistently observed a significant benefit of surgery among indicated patients.
Comparisons of discectomy with an active control may help limit expectation bias and crossover rates. Accordingly, a Cochrane Systematic Review of RCTs on surgical interventions for LDH also compared discectomy with chemonucleolysis.36 Meta-analyses in the Cochrane Review demonstrated strong evidence for superiority of chemonucleolysis over placebo as well as strong evidence for the superiority of discectomy over chemonucleolysis, which also suggests that discectomy is more effective than placebo.36
Endoscopic versus open discectomy
Endoscopic discectomy is a minimally invasive technique designed to reduce tissue trauma compared to open discectomy.37,38 As with an open procedure, an endoscopic approach allows for full access to the herniated disc under direct visualization using an endoscope. Advantages of endoscopic compared to open discectomy include reduced incision size and soft-tissue damage, which may be associated with faster recovery.39 The transforaminal endoscopic approach obviates the need for removal of laminar bone and ligamentum flavum, resulting in less alteration of the anatomy. Disadvantages include an unfamiliar approach to most surgeons and a learning curve, making this approach technically challenging for many surgeons.40
Clinical studies and meta-analyses have shown that patients treated with endoscopic discectomy experience similar improvements in function and symptom relief when compared to traditional open discectomy or microdiscectomy.38,41-45 Ruetten et al. performed a seminal 200-patient RCT, with randomization to either conventional micro-discectomy (n=100) or full-endoscopic discectomy (transforaminal, n = 41; interlaminar, n=59).38 There were no serious complications in either group, but the micro-discectomy group had a significantly higher rate of non-serious complications, such as postoperative bleeding or delayed wound healing (P<0.05). Recurrent herniation rates were also similar between the groups, but the rate of recurrence was significantly higher among patients who had a large vs. small annular defect, regardless of treatment type (P<0.01). Clinical outcomes, such as leg pain and disability scores, were significantly improved in both groups through 2 years follow-up (89% follow-up rate), with no significant differences between groups. However, the endoscopic group reported significantly less postoperative pain, less pain medication use, and fewer postoperative work disability days (25 days vs. 49 days) than the micro-discectomy group (P<0.01).
Gibson et al. performed a prospective RCT comparing 70 patients treated with transforaminal endoscopic discectomy (TED) versus 70 patients treated with microdiscectomy and observed similar outcomes for improvements in pain and disability between the two groups.42 However, patients in the TED group had a shorter mean hospital stay (0.7 versus 1.4 nights; P<0.001), suggesting a faster recovery time. Zhang et al. reported results from a systematic review of 9 studies (1,527 patients) comparing microdiscectomy with TED.45 Results from the meta-analysis suggested that there were no significant differences in visual analog scale (VAS) leg pain, recovery of function following surgery, operative time, or incidence of complications. Similar to the study by Gibson et al., endoscopic discectomy significantly reduced the length of hospital stay (P<0.001).
Prevention of recurrent LDH
Although primary discectomy is an effective treatment for LDH with radiculopathy, a small proportion of patients (5-15%) experience symptomatic recurrent LDH (rLDH)19,46,47 and 4-6% undergo revision surgery within 2 years.48-50 Techniques for management of recurrent herniation are dependent upon the specific patient indications and the surgeon’s experience, on a case-by-case basis. Regardless of the reoperation strategy, clinical evidence has shown that patients who undergo revision surgery for symptomatic rLDH have worse clinical outcomes (e.g. pain and disability) compared to patients who do not reherniate following primary discectomy.51-57 Abdu et al. observed that 9.1% of the 810 SPORT patients treated with discectomy experienced rLDH and revision surgery during the 8-year follow-up period.52 Compared to patients that did not reherniate, those with rLDH and revision surgery had significantly worse SF-36 bodily pain, ODI, and sciatica bothersomeness scores at 4 years follow-up (P≤0.001 for all metrics). Similarly, a study of patient-reported outcomes from a large registry (Swespine) demonstrated that patients who underwent revision surgery for rLDH experienced significantly less improvement in VAS leg pain, VAS back pain, ODI, and EQ-5D health status compared to the primary LDH group (P≤0.001 for all metrics).53
Patient factors that may increase the risk of rLDH include younger age, male gender, work that requires lifting, worse baseline pain and disability, lack of motor or sensory deficit, smoking, and diabetes.46,52,58 An important risk factor for symptomatic rLDH that may be addressable by the surgeon is a large annular defect (≥ 6 mm wide). A recent (2018) meta-analysis reported that a large annular defect increases the odds of a patient experiencing symptom recurrence or revision surgery by a factor of 2.5 or 2.3, respectively.20 There is an unmet medical need to address this elevated risk in patients with large annular defects and avoid the worse outcomes associated with revision.59 Several devices have been designed to help repair large annular defects, including soft tissue suture kits and bone-anchored annular closure devices (ACD). While discectomy is intended to alleviate patient symptoms by decompressing the impinged neural structures, annular repair is intended to reduce the risk of recurrence and revision, thereby sustaining the clinical benefits of the primary discectomy.
Currently, clinical evidence exists for two annular repair methods: 1) closure of the annular defect using a soft tissue suture kit and 2) implantation of a bone-anchored ACD. Bailey et al. described a 750-patient RCT to study annular repair using a soft tissue suture system that was commercially available at the time of the study.60 Patients were randomized 2:1 to annular repair or no annular repair (control) after discectomy. Included subjects were candidates for 1- or 2-level lumbar discectomy, had persistent and predominant leg pain (VAS leg > 4 / 10), and were not responsive to 6 weeks of nonsurgical care. No inclusion or exclusion criteria specified the size of the annular defect. The authors reported that annulotomy was performed in 77% of patients, producing annular defects with a mean size of 4.75 ± 2.0 mm. Assuming a normal distribution, 73% of annulotomy patients had a defect size less than 6 mm, which is commonly described as the threshold for “large” defects.20,59 There were no significant differences in rates of adverse events or serious adverse events between the two groups. Clinical outcomes of leg pain, back pain, disability, and physical function were also similar between the groups over 2 years of follow-up. While there was a trend of reduced rLDH-related reoperations over 2 years in the annular repair group, the results were not statistically significant (9.7% vs. 11.2%; P=0.56). The marginal reductions in reoperation rate may have been related to the fact that most patients did not have a large annular defect. In the subgroup with predominant leg pain, the rate of rLDH-related reoperations was significantly reduced in the annular repair group at 3 months (1.0% vs. 5.9%; P=0.019) and 6 months (2.0% vs. 6.9%; P=0.046) follow-up, but not at 2 years (6.7% vs. 12.1%; P=0.13).
Several clinical studies, including 2 RCTs and a prospective comparative cohort study,61-63 have shown that bone-anchored ACD implantation reduces the risk of symptomatic rLDH and reoperation compared with discectomy alone (Table 2). Four additional prospective cohort or registry studies have also observed positive outcomes and low revision rates with this treatment technique.64-68 One multicenter RCT analyzed 550 patients with large (≥ 6 mm wide) annular defects who were randomized 1:1 to treatment with discectomy alone (n=278) or discectomy plus a bone-anchored ACD (n=272).62,69 The ACD group experienced a significantly lower rate of symptomatic rLDH (12% vs 25%; P<0.001) and related revision surgeries (5% vs 13%; P=0.001) compared to discectomy alone over 2 years of follow-up.62 At 3 and 4 years follow-up, the ACD group also experienced significantly greater improvement in pain, disability, and health-related quality of life scores compared to discectomy alone (P<0.05).70,71 Based on Kaplan-Meier survival analysis, the incidence rates continued to be lower in the ACD group over a 5-year follow-up for symptomatic index-level rLDH (21.8% vs. 32.8%; P<0.001) and index-level reoperation (16.2% vs. 22.7%; P=0.03).72
Table 2. Summary of clinical studies on bone-anchored annular closure vs. discectomy alone
Study Citation
Patient Population Study Design
Sample Size
Follow-up Time
Results
Klassen et al. 2017 69 * Radiculopathy + imaging confirmed LDH + failed ≥ 6 wks of NS care; large annular defect (≥6 mm); mean ages were 43 (22-71) (AC) and 44 (23-74) (D) yrs RCT D: 278 AC: 272
90 d Significantly lower rate of all-cause SAEs (9.7% vs 16.3%; P=0.056) and device- or surgery-related SAEs (4.5% vs 10.2%; P=0.02) with AC compared to D at 90 d follow-up; by 90 d, reoperation rate was 5.4% with D and 1.9% with AC (P=0.03); in the AC group, device dislocation occurred in 0.7% of AC patients Thomé et al 2018 62 * 2 yrs Frequency of symptomatic index-level rLDH (12% vs 25%; P<0.001), composite success index (27% vs 18%; P=0.02), and device- or procedure-related SAEs (7% vs 17%; P=0.001) significantly improved with AC compared to D; reoperation rate was 13% in D and 5% in AC groups (P=0.001); device migration and mesh migration occurred in 1.1% and 1.5% of AC patients, respectively Kienzler et al. 2019 70 * 3 yrs Symptomatic index-level rLDH rate (14.8% vs 29.5%; P<0.001), reoperation rate (11% vs 19.3%; P=0.007), leg pain (21 vs 30; P<0.01), back pain (23 vs 30; P=0.01), ODI (18 vs 23; P=0.02), physical health component (47 vs 44; P<0.01), and mental health component (52 vs 49; P<0.01) scores were significantly improved with AC compared to D; reoperation rate was 19.3% with D and 11% with AC (P=0.007); all-cause SAE rate was reduced in AC group (10.7% vs 18.7%; P=0.008); device-related deficiencies and fracture occurred in 4% and 0.4% of AC patients, respectively Nanda et al. 2019 71 * 4 yrs Over 4 years, reoperation rate was significantly lower in AC group (14.4% vs. 21.1%; P=0.03). Partial or complete device removal in 23 reoperations. No reoperations were related to endplate changes. Magnitude of improvement was greater in AC patients for leg pain (P=0.04), ODI (P=0.04), and mental health component scores (P=0.02). Cho et al. 2019 63 Radiculopathy + radiographically confirmed LDH; mean ages 41 ± 11 (AC) and 43 ± 12 (D) yrs RCT D: 30 AC: 30
2 yrs Significantly greater disc height maintenance (86.3% vs 79.2%; P=0.04) in AC group compared to D group; rate of rLDH was 20% in D and 3.3% in AC groups (P=0.04); no instances of device migration, loosening, or fracture occurred in AC group Ardeshiri et al. 2019 68 Symptomatic, MRI-confirmed LDH at single level; disc height ≥ 5 mm; annular defect ≥6 mm; mean age 45 (23-82) yrs Prospective Cohort AC: 75 2 yrs rLDH rate was 1.4% (1/75). Three reoperations required (4%): rLDH, epidural infection, and device dislocation. One intra-operative, implant-associated dural tear, which did not result in post-operative complications. Improper implantation of one device in one patient, but no associated adverse events noted. Sanginov et al. 2018 67 Symptomatic LDH confirmed by CT or MRI + failed 6 wks of NS care. Mean age of 38 (17-63) yrs Prospective Cohort AC: 120 5 yrs One ipsilateral (0.8%) and one contralateral (0.8%) rLDH, each of which were reoperated. Other reoperations included 3 fusions for segmental instability and treatment of 1 epidural hematoma. Endplate changes were present in 20 patients (16.7%) at baseline and 42 (35%) within 5 yrs of follow-up. No significant differences in pain or function scores between patients with or without endplate changes. Kursumovic et al. 2017 66 Radiculopathy + radiographically confirmed LDH; 90% with annular defect ≥6 mm; mean age 46 (18-75) yrs Real-world registry analysis AC: 171 15 mo Mean follow-up of 15 mo (1-72 mo); 74% had 12 mo or later follow-up. Six (3.5%) reherniations, with four (2.3%) of those six requiring reoperation. A total of 22 reoperations were performed across 12 subjects (7%), which included 5 fusions, 5 re-discectomies, 4 wound revisions, 3 decompressions, 2 spinal cord stimulators, and 3 unknowns. Device-related complications in 15 patients (8.8%), 2 of which required revision surgery. Vukas et al. 2013 61 Radiculopathy + failed ≥ 6 wks of NS care + preoperative MRI; mean ages 38 (AC) and 41 (D) (18-70 for all) yrs Prospective Comparative Cohorts
D: 72 AC: 30
2 yrs Improved leg pain (8.9 vs 21.2; P=0.005), back pain (10.5 vs 19.1; P=0.27), and disability (11.6 vs 19.8; P=0.08) with AC compared to D at 2 yrs follow-up; symptomatic reherniation rate was 6.9% in D and 0% in AC groups; patients were enrolled non-concurrently; no instances of device-related AEs Lequin et al. 2012 64 Sciatica with MRI-confirmed LDH + failed
≥ 6 wks of NS care. Mean age 42 ± 11 yrs. Mean defect width 7.8 mmProspective Cohort AC: 45 1 yr Symptomatic rLDH in 1/41 (2.4%) of patients, which also required a reoperation. Three total reoperations (6.7%): ipsilateral rLDH, contralateral rLDH, and scar tissue management. No evidence of heterotopic ossification or spontaneous fusion. Zero device complications observed. Abbreviations: D, discectomy group; AC, annular closure; NS, nonsurgical; RCT, randomized controlled trial; MRI, magnetic resonance imaging; LDH, lumbar disc herniation; rLDH, recurrent lumbar disc herniation; AE, adverse events; SAE, serious adverse events; yrs, years; y, year; mo, months; d, days * These studies report 90 d, 2 yrs, 3 yrs, and 4 yrs follow-up timepoints and outcomes of the same RCT.
According to the Food and Drug Administration’s (FDA) Summary of Safety and Effectiveness Data (SSED) related to this device and RCT, there were 34 (12.7%) device deficiencies comprised of 5 anchor migrations and 29 events related to the occlusion component.72 Approximately half of the deficiencies were asymptomatic and 12 (4.5%) of those deficiencies were related to a serious adverse event (SAE). Despite these device-related complications, the ACD group experienced significantly fewer device- or procedure-related SAEs over a 5-year time period (P=0.037) due to the reduction in symptomatic rLDH rates.72 The SSED also described the observation of radiographic endplate lesions (EPL) on CT images at a significantly higher rate in the ACD group (89% vs. 41%; P<0.001). However, the size of 90% of the EPLs in both groups stabilized over the 3- to 5-year observation period. Further, in the ACD group, no negative clinical outcomes were associated with the EPLs, based on patient-reported outcomes, reoperations, or SAEs. Current evidence suggests that the EPLs do not impact device function or clinical outcomes at 5 years follow-up and an ongoing study will continue to monitor safety.72
Cho et al. reported the results from a separate 60-patient RCT, where 30 patients received discectomy and 30 received discectomy plus a bone-anchored ACD.63 By 2 years, the ACD group had a significantly lower rate of rLDH (3.3% vs 20%; P=0.04) compared to discectomy alone. Disc height was also better maintained at 2 years follow-up in the bone-anchored ACD group compared to control (86.3% vs. 79.2%; P=0.04). Both groups experienced significant improvements in all patient-reported outcomes related to leg and back pain, ODI, and quality of life (P<0.001). The authors reported that no complications, including device migration or loosening, were observed in any patients within the 2-year follow-up.63
In addition to the two RCTs, four prospective cohort studies and one retrospective analysis of a real-world treatment registry were identified (513 patients total).61,64,66-68 Each of these studies described positive treatment results with a bone-anchored ACD, with rLDH rates ranging from 0% to 3.5% and reoperation rates ranging from 0% to 7.3% over a 1- to 5-year follow-up period. Device-related adverse events occurred at similar rates to the RCT (2% – 8.8%). The current evidence for bone-anchored annular closure is derived from 7 unique studies and 1,123 patients (ACD: n=743; Control: n=380). Collectively, this evidence indicates a positive benefit-risk ratio for lumbar discectomy patients with large annular defects, based on significant reductions in symptomatic rLDH and revision surgery in an at-risk population.
Cost-effectiveness
In addition to the clinical benefits, primary discectomy has been shown to be cost-effective relative to nonsurgical care. A 1996 study found that discectomy increases quality-adjusted life years (QALY) by 0.43 over 10 years at a non-discounted cost of $29,200 per QALY.73 More recent (2008 and 2011) studies from Tosteson et al. reported that the costs for discectomy per QALY ranged from $34,355 to $69,403 over 2 years follow-up and $20,600 per QALY through 4 years.74,75 Furthermore, data from the National Health Interview Survey show that patients treated with discectomy return to work faster, miss fewer workdays, have increased average earnings, and the increased earnings offset the cost of surgery.76 When also considering the effects on employee productivity, surgical discectomy may yield a net societal cost savings.76
Compelling evidence has shown that primary discectomy is a safe and effective treatment for indicated patients; yet, a minority of patients experience recurring symptoms, and some undergo revision surgery. Reoperation adds over $44,000 per patient in direct healthcare costs over 4 years following discectomy.77 With approximately 500,000 discectomies per year in the United States,78 it is important from both economic and socioeconomic standpoints to identify evidence-based treatment strategies that help reduce the burden associated with revision surgeries after discectomy.
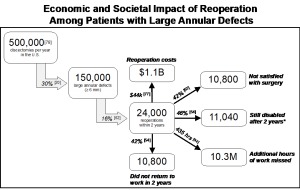
Among at-risk patients with large annular defects, we estimate there could be 24,000 reoperations over any two-year period in the United States, which are associated with a substantial economic and societal impact (Figure 1). Annular repair has reduced the rLDH-related reoperation rate by over 60% in a two-year time period,62 which should reduce the direct healthcare costs, allow more patients to return to work and stay at work, and avoid the significant morbidity associated with reoperation. Based on RCT data, a bone-anchored ACD was found to be cost-effective at $6,030 per QALY over 2 years follow-up, which is well below the standard willingness-to-pay thresholds.79 When the societal costs of missed work and lower productivity were also considered, the incremental cost-effectiveness ratio of the bone-anchored ACD became negative, which indicates “economic dominance” – improved outcomes at a lower cost – compared to discectomy alone.79
Figure 1. Estimated impact of rLDH-related reoperation among patients with large annular defects.
Coverage Rationale
Nonsurgical management is generally the first line of treatment for patients with LDH and radiculopathy. For patients with symptomatic LDH that fail to improve after 6 weeks of nonsurgical care, or have severe symptoms that necessitate earlier intervention, large bodies of evidence indicate that discectomy is medically necessary and provides superior outcomes compared to continuing nonsurgical care. Each discectomy approach (open, microtubular, or endoscopic) has demonstrated similar clinical benefits and is appropriate for the surgical treatment of LDH with radiculopathy. The current high-level clinical evidence supports discectomy (open, microtubular, or endoscopic discectomy) as a medically necessary procedure for the treatment of LDH with radiculopathy in indicated patients.
In patients undergoing primary discectomy with a large (≥ 6 mm wide) annular defect, annular repair or closure is intended to control the risk of recurring symptoms and reoperation. Currently, the only annular repair technique supported by high-level clinical evidence is bone-anchored annular closure. A bone-anchored ACD is FDA-approved and indicated for patients with large annular defects. Adequate evidence is currently not available to support suturing of the annulus as an effective repair technique. The current clinical evidence supports bone-anchored annular closure for indicated patients to improve the probability of avoiding rLDH and sustaining the treatment benefits of discectomy.
Indications/ Limitations of Coverage
Patients who have the following clinical indications may be eligible for discectomy and coverage:
- Clinical signs and symptoms associated with LDH
- Imaging confirmation of LDH consistent with clinical findings
- Failure to improve after 6 weeks of conservative care
Clinical indications for annular closure and coverage include (based on the FDA-approved indications):
- Patient is indicated for a primary discectomy due to a posterior or posterolateral herniation
- Discectomy will be performed at a single level that includes L4-L5 or L5-S1
- The annular defect is large (between 4-6 mm tall and between 6-10 mm wide) after completion of the discectomy procedure
Coding
Procedural (CPT) Coding
The American Medical Association recommends that posterior/posterolateral and/or revision lumbar discectomy be coded using Current Procedural Terminology (CPT) that are listed below. For purposes of CPT coding, the primary approach and visualization define the service. Unless otherwise specified, lumbar discectomy is presumed open (which includes microtubular approach).
Definitions:
Direct visualization: Light-based visualization can [only] be performed by eye, or with surgical loupes, microscope, or endoscope.
Open: Spinal procedures with continuous direct [light-based] visualization of the spine through a surgical opening.
Endoscopic: Spinal procedures performed with continuous direct [light-based] visualization of the spine through a surgical opening.
CPT Codes:
63030: Laminotomy (hemilaminectomy) with decompression of nerve root(s) including partial facetectomy, foraminotomy, and/or excision of herniated intervertebral disc; 1 interspace, lumbar (For bilateral procedure, report 63030 with modifier 50)
63035: each additional interspace, lumbar
(Use 63035 in conjunction with 63030)
(For bilateral procedure, report 63035 with modifier 50)
63042: Laminotomy (hemilaminectomy) with decompression of nerve root(s), including partial facectomy, foraminotomy, and/or excision of herniated intervertebral, reexploration, single interspace, lumbar
(For bilateral procedure, report 63042 with modifier 50)
63044: each additional lumbar interspace (List separately in addition to code for primary procedure) (Use 63044 in conjunction with 63042)
63056: Transpedicular approach with decompression of spinal cord, equina, and/or nerve root(s) single segment; lumbar (including transfacet, or lateral extraforaminal approach) (eg, far lateral herniated intervertebral disc)
63057: each additional segment, lumbar (List separately in addition to code for primary procedure) (Use 63057 in conjunction with 63056)
62380: Endoscopic decompression of spinal cord, nerve root(s) including laminotomy, partial facectomy, foraminotomy, discectomy and/or excision of herniated intervertebral disc, 1 interspace, lumbar (For bilateral procedure, report 62380 with modifier 50)
(Effective Date: January 2017)
22899: Unlisted procedure, spine
Physicians may report CPT 22899 for implantation of the bone-anchored annular closure device in addition to the appropriate CPT code for the discectomy procedure (e.g., 63030)
Diagnostic (ICD10-CM) Coding
Table 3. Diagnosis Codes (ICD-CM)
Diagnosis Code Code Descriptor M51 Thoracic, thoracolumbar, and lumbosacral intervertebral disc disorders M51.0 with myelopathy M51.1 with radiculopathy M51.2 Other thoracic, thoracolumbar and lumbosacral intervertebral disc displacement M51.3 Other thoracic, thoracolumbar and lumbosacral intervertebral disc degeneration M51.8 Other thoracic, thoracolumbar and lumbosacral intervertebral disc disorders M51.9 Unspecified thoracic, thoracolumbar and lumbosacral intervertebral disc disorder M48.00 Spinal stenosis, site unspecified M48.05 Spinal stenosis, thoracolumbar region M48.06 Spinal stenosis, lumbar region M48.07 Spinal stenosis, lumbosacral region M48.08 Spinal stenosis, sacral and sacrococcygeal region M47.1 Other spondylosis with myelopathy M47.2 Other spondylosis with radiculopathy M47.16 Other spondylosis with myelopathy, lumbar region M47.81 Spondylosis without myelopathy or radiculopathy M47.89 Other spondylosis M47.9 Spondylosis, unspecified M43.00 Spondylolysis, site unspecified M43.05 Spondylolysis, thoracolumbar region M43.06 Spondylolysis, lumbar region M43.07 Spondylolysis, lumbosacral region M43.08 Spondylolysis, sacral and sacrococcygeal region M43.09 Spondylolysis, multiple sites in spine M54.5 Low back pain M54.8 Other dorsalgia M54.9 Dorsalgia, unspecified Hospital Outpatient/ASC Coding
Medicare created a new Healthcare Common Procedure Coding System (HCPCS) code that specifically includes a bone-anchored annular closure device that hospital outpatient departments and ambulatory surgical centers (ASCs) should report for the procedure:
C9757: Laminotomy (hemilaminectomy), with decompression of nerve root(s), including partial facetectomy, foraminotomy and excision of herniated intervertebral disc, and repair of annular defect with implantation of bone anchored annular closure device, including annular defect measurement, alignment and sizing assessment, and image guidance; 1 interspace, lumbar
(Effective Date: January 2020)
Documentation Requirements
1) Radiculopathy confirmed on history and physical examination.
AND
2) EITHER
– Disabling leg or back pain refractory to 6 weeks of conservative care including any one of the following: time, physician structured exercise regimen, lumbar epidural therapy, or physical/chiropractic therapy
OR
– Progressive neurologic deficit
AND
3) Level appropriate documentation of nerve root compression on imaging [MRI]/[CT, preferably with Myelogram] and/or nerve conduction velocity/electromyogram (NCV/EMG).
Conclusion
Discectomy is a safe and effective procedure for patients with primary LDH and radiculopathy who do not respond to nonsurgical care or have severe and deteriorating symptoms. The current evidence is consistently in favor of discectomy, rather than continuing nonsurgical care, for greater and/or faster alleviation of pain, reduction of disability, and higher patient satisfaction. Clinical evidence indicates that microtubular discectomy and endoscopic discectomy result in reduced incision size, soft tissue damage, and hospital stay and yield similar improvements in symptoms compared to open discectomy. Currently, evidence-based clinical recommendations for annular repair after discectomy do not exist. Patients who are indicated for discectomy and have a large annular defect are exposed to a greater risk of symptom recurrence and revision surgery and may benefit from annular repair. Current level I evidence demonstrates that, in appropriately selected patient populations, implantation of a bone-anchored ACD reduces the risk of symptom recurrence and revision surgery compared to discectomy alone. The current evidence supports the use of discectomy as well as consideration for bone-anchored annular repair for the treatment of indicated patients with primary symptomatic LDH who may otherwise be at higher risk for recurrent disc herniation. [This ISASS Policy addresses only LDH with radiculopathy that is clinically appropriate for direct visualization (open, microtubular, or endoscopic discectomy); the particular primary posterior/posterolateral surgical procedural approach is the purview of the surgeon.]
Corresponding Author:
Morgan Lorio, MD, FACS, Advanced Orthopedics, 499 E. Central Parkway, Altamonte Springs, FL 32701 Phone: (407) 960-1717; Email: mloriomd@gmail.com
References
- Kreiner DS, Hwang SW, Easa JE, et al. An evidence-based clinical guideline for the diagnosis and treatment of lumbar disc herniation with radiculopathy. Spine J. 2014;14(1):180-191.
- Fardon DF, Milette PC, Combined Task Forces of the North American Spine Society ASoSR, American Society of N. Nomenclature and classification of lumbar disc pathology. Recommendations of the Combined task Forces of the North American Spine Society, American Society of Spine Radiology, and American Society of Neuroradiology. Spine (Phila Pa 1976). 2001;26(5):E93-E113.
- Frymoyer JW. Back pain and sciatica. N Engl J Med. 1988;318(5):291-300.
- Amin RM, Andrade NS, Neuman BJ. Lumbar Disc Herniation. Curr Rev Musculoskelet Med. 2017;10(4):507-516.
- Koes BW, van Tulder MW, Peul WC. Diagnosis and treatment of sciatica. BMJ. 2007;334(7607):1313-1317.
- Tawa N, Rhoda A, Diener I. Accuracy of clinical neurological examination in diagnosing lumbo-sacral radiculopathy: a systematic literature review. BMC Musculoskelet Disord. 2017;18(1):93.
- Janssen ME, Bertrand SL, Joe C, Levine MI. Lumbar herniated disk disease: comparison of MRI, myelography, and post-myelographic CT scan with surgical findings. Orthopedics. 1994;17(2):121-127.
- Mysliwiec LW, Cholewicki J, Winkelpleck MD, Eis GP. MSU classification for herniated lumbar discs on MRI: toward developing objective criteria for surgical selection. Eur Spine J. 2010;19(7):1087-1093.
- Wong JJ, Cote P, Sutton DA, et al. Clinical practice guidelines for the noninvasive management of low back pain: A systematic review by the Ontario Protocol for Traffic Injury Management (OPTIMa) Collaboration. Eur J Pain. 2017;21(2):201-216.
- Dagenais S, Tricco AC, Haldeman S. Synthesis of recommendations for the assessment and management of low back pain from recent clinical practice guidelines. Spine J. 2010;10(6):514-529.
- Peul WC, van Houwelingen HC, van den Hout WB, et al. Surgery versus prolonged conservative treatment for sciatica. N Engl J Med. 2007;356(22):2245-2256.
- Osterman H, Seitsalo S, Karppinen J, Malmivaara A. Effectiveness of microdiscectomy for lumbar disc herniation: a randomized controlled trial with 2 years of follow-up. Spine (Phila Pa 1976). 2006;31(21):2409-2414.
- Buttermann GR. Treatment of lumbar disc herniation: epidural steroid injection compared with discectomy. A prospective, randomized study. J Bone Joint Surg Am. 2004;86(4):670-679.
- Weinstein JN, Tosteson TD, Lurie JD, et al. Surgical vs nonoperative treatment for lumbar disk herniation: the Spine Patient Outcomes Research Trial (SPORT): a randomized trial. JAMA. 2006;296(20):2441-2450.
- Gadjradj PS, Arts MP, van Tulder MW, Rietdijk WJR, Peul WC, Harhangi BS. Management of Symptomatic Lumbar Disk Herniation: An International Perspective. Spine (Phila Pa 1976). 2017;42(23):1826-1834.
- Health Technology Clinical Committee Findings and Decision. Paper presented at: Surgery for lumbar radiculopathy/sciatica; May 18, 2018; SeaTac Conference Center, Seattle, Washington.
- Surgery for Lumbar Radiculopathy/Sciatica: Final evidence report. In:2018.
- Fjeld OR, Grovle L, Helgeland J, et al. Complications, reoperations, readmissions, and length of hospital stay in 34 639 surgical cases of lumbar disc herniation. Bone Joint J. 2019;101-B(4):470-477.
- Carragee EJ, Han MY, Suen PW, Kim D. Clinical outcomes after lumbar discectomy for sciatica: the effects of fragment type and anular competence. J Bone Joint Surg Am. 2003;85(1):102-108.
- Miller LE, McGirt MJ, Garfin SR, Bono CM. Association of Annular Defect Width After Lumbar Discectomy With Risk of Symptom Recurrence and Reoperation: Systematic Review and Meta-analysis of Comparative Studies. Spine (Phila Pa 1976). 2018;43(5):E308-E315.
- Atlas SJ, Deyo RA, Keller RB, et al. The Maine Lumbar Spine Study, Part II. 1-year outcomes of surgical and nonsurgical management of sciatica. Spine (Phila Pa 1976). 1996;21(15):1777-1786.
- Gugliotta M, da Costa BR, Dabis E, et al. Surgical versus conservative treatment for lumbar disc herniation: a prospective cohort study. BMJ Open. 2016;6(12):e012938.
- Weinstein JN, Lurie JD, Tosteson TD, et al. Surgical vs nonoperative treatment for lumbar disk herniation: the Spine Patient Outcomes Research Trial (SPORT) observational cohort. JAMA. 2006;296(20):2451-2459.
- McMorland G, Suter E, Casha S, du Plessis SJ, Hurlbert RJ. Manipulation or microdiskectomy for sciatica? A prospective randomized clinical study. J Manipulative Physiol Ther. 2010;33(8):576-584.
- Thomas KC, Fisher CG, Boyd M, Bishop P, Wing P, Dvorak MF. Outcome evaluation of surgical and nonsurgical management of lumbar disc protrusion causing radiculopathy. Spine (Phila Pa 1976). 2007;32(13):1414-1422.
- Atlas SJ, Keller RB, Chang Y, Deyo RA, Singer DE. Surgical and nonsurgical management of sciatica secondary to a lumbar disc herniation: five-year outcomes from the Maine Lumbar Spine Study. Spine (Phila Pa 1976). 2001;26(10):1179-1187.
- Atlas SJ, Keller RB, Wu YA, Deyo RA, Singer DE. Long-term outcomes of surgical and nonsurgical management of lumbar spinal stenosis: 8 to 10 year results from the maine lumbar spine study. Spine (Phila Pa 1976). 2005;30(8):936-943.
- Weinstein JN, Lurie JD, Tosteson TD, et al. Surgical versus nonoperative treatment for lumbar disc herniation: four-year results for the Spine Patient Outcomes Research Trial (SPORT). Spine (Phila Pa 1976). 2008;33(25):2789-2800.
- Lurie JD, Tosteson TD, Tosteson AN, et al. Surgical versus nonoperative treatment for lumbar disc herniation: eight-year results for the spine patient outcomes research trial. Spine (Phila Pa 1976). 2014;39(1):3-16.
- McCoy CE. Understanding the Intention-to-treat Principle in Randomized Controlled Trials. West J Emerg Med. 2017;18(6):1075-1078.
- Hernan MA, Hernandez-Diaz S. Beyond the intention-to-treat in comparative effectiveness research. Clin Trials. 2012;9(1):48-55.
- Pearson A, Lurie J, Tosteson T, et al. Who should have surgery for an intervertebral disc herniation? Comparative effectiveness evidence from the spine patient outcomes research trial. Spine (Phila Pa 1976). 2012;37(2):140-149.
- Kerr D, Zhao W, Lurie JD. What Are Long-term Predictors of Outcomes for Lumbar Disc Herniation? A Randomized and Observational Study. Clin Orthop Relat Res. 2015;473(6):1920-1930.
- Arts MP, Kursumovic A, Miller LE, et al. Comparison of treatments for lumbar disc herniation: Systematic review with network meta-analysis. Medicine (Baltimore). 2019;98(7):e14410.
- Lurie JD, Henderson ER, McDonough CM, et al. Effect of Expectations on Treatment Outcome for Lumbar Intervertebral Disc Herniation. Spine (Phila Pa 1976). 2016;41(9):803-809.
- Gibson JN, Waddell G. Surgical interventions for lumbar disc prolapse: updated Cochrane Review. Spine (Phila Pa 1976). 2007;32(16):1735-1747.
- Ahn Y. Endoscopic spine discectomy: indications and outcomes. Int Orthop. 2019;43(4):909-916.
- Ruetten S, Komp M, Merk H, Godolias G. Full-endoscopic interlaminar and transforaminal lumbar discectomy versus conventional microsurgical technique: a prospective, randomized, controlled study. Spine (Phila Pa 1976). 2008;33(9):931-939.
- Rasouli MR, Rahimi-Movaghar V, Shokraneh F, Moradi-Lakeh M, Chou R. Minimally invasive discectomy versus microdiscectomy/open discectomy for symptomatic lumbar disc herniation. Cochrane Database Syst Rev. 2014(9):CD010328.
- Butler AJ, Alam M, Wiley K, Ghasem A, Rush IA, Wang JC. Endoscopic Lumbar Surgery: The State of the Art in 2019. Neurospine. 2019;16(1):15-23.
- Hermantin FU, Peters T, Quartararo L, Kambin P. A prospective, randomized study comparing the results of open discectomy with those of video-assisted arthroscopic microdiscectomy. J Bone Joint Surg Am. 1999;81(7):958-965.
- Gibson JNA, Subramanian AS, Scott CEH. A randomised controlled trial of transforaminal endoscopic discectomy vs microdiscectomy. Eur Spine J. 2017;26(3):847-856.
- Lee SH, Chung SE, Ahn Y, Kim TH, Park JY, Shin SW. Comparative radiologic evaluation of percutaneous endoscopic lumbar discectomy and open microdiscectomy: a matched cohort analysis. Mt Sinai J Med. 2006;73(5):795-801.
- Ahn SS, Kim SH, Kim DW, Lee BH. Comparison of Outcomes of Percutaneous Endoscopic Lumbar Discectomy and Open Lumbar Microdiscectomy for Young Adults: A Retrospective Matched Cohort Study. World Neurosurg. 2016;86:250-258.
- Zhang B, Liu S, Liu J, et al. Transforaminal endoscopic discectomy versus conventional microdiscectomy for lumbar discherniation: a systematic review and meta-analysis. J Orthop Surg Res. 2018;13(1):169.
- Huang W, Han Z, Liu J, Yu L, Yu X. Risk Factors for Recurrent Lumbar Disc Herniation: A Systematic Review and Meta-Analysis. Medicine (Baltimore). 2016;95(2):e2378.
- Tanavalee C, Limthongkul W, Yingsakmongkol W, Luksanapruksa P, Singhatanadgige W. A comparison between repeat discectomy versus fusion for the treatment of recurrent lumbar disc herniation: Systematic review and meta-analysis. J Clin Neurosci. 2019.
- Parker SL, Mendenhall SK, Godil SS, et al. Incidence of Low Back Pain After Lumbar Discectomy for Herniated Disc and Its Effect on Patient-reported Outcomes. Clin Orthop Relat Res. 2015;473(6):1988-1999.
- Virk SS, Diwan A, Phillips FM, Sandhu H, Khan SN. What is the Rate of Revision Discectomies After Primary Discectomy on a National Scale? Clin Orthop Relat Res. 2017;475(11):2752-2762.
- Elkan P, Lagerback T, Moller H, Gerdhem P. Response rate does not affect patient-reported outcome after lumbar discectomy. Eur Spine J. 2018;27(7):1538-1546.
- Vik A, Zwart JA, Hulleberg G, Nygaard OP. Eight year outcome after surgery for lumbar disc herniation: a comparison of reoperated and not reoperated patients. Acta Neurochir (Wien). 2001;143(6):607-610; discussion 610-611.
- Abdu RW, Abdu WA, Pearson AM, Zhao W, Lurie JD, Weinstein JN. Reoperation for Recurrent Intervertebral Disc Herniation in the Spine Patient Outcomes Research Trial: Analysis of Rate, Risk Factors, and Outcome. Spine (Phila Pa 1976). 2017;42(14):1106-1114.
- Fritzell P, Knutsson B, Sanden B, Stromqvist B, Hagg O. Recurrent Versus Primary Lumbar Disc Herniation Surgery: Patient-reported Outcomes in the Swedish Spine Register Swespine. Clin Orthop Relat Res. 2015;473(6):1978-1984.
- Klassen PD, Hsu WK, Martens F, et al. Post-lumbar discectomy reoperations that are associated with poor clinical and socioeconomic outcomes can be reduced through use of a novel annular closure device: results from a 2-year randomized controlled trial. Clinicoecon Outcomes Res. 2018;10:349-357.
- Lebow RL, Adogwa O, Parker SL, Sharma A, Cheng J, McGirt MJ. Asymptomatic same-site recurrent disc herniation after lumbar discectomy: results of a prospective longitudinal study with 2-year serial imaging. Spine (Phila Pa 1976). 2011;36(25):2147-2151.
- Leven D, Passias PG, Errico TJ, et al. Risk Factors for Reoperation in Patients Treated Surgically for Intervertebral Disc Herniation: A Subanalysis of Eight-Year SPORT Data. J Bone Joint Surg Am. 2015;97(16):1316-1325.
- Nolte M, Basques BA, Louie PK, et al. Patients Undergoing Revision Microdiskectomy for Recurrent Lumbar Disk Herniation Experience Worse Clinical Outcomes and More Revision Surgeries Compared With Patients Undergoing a Primary Microdiskectomy. J Am Acad Orthop Surg. 2019.
- Osterman H, Sund R, Seitsalo S, Keskimaki I. Risk of multiple reoperations after lumbar discectomy: a population-based study. Spine (Phila Pa 1976). 2003;28(6):621-627.
- Ammerman J, Watters WC, Inzana JA, Carragee G, Groff MW. Closing the Treatment Gap for Lumbar Disc Herniation Patients with Large Annular Defects: A Systematic Review of Techniques and Outcomes in this High-risk Population. Cureus. 2019;11(5):e4613.
- Bailey A, Araghi A, Blumenthal S, Huffmon GV, Anular Repair Clinical Study G. Prospective, multicenter, randomized, controlled study of anular repair in lumbar discectomy: two-year follow-up. Spine (Phila Pa 1976). 2013;38(14):1161-1169.
- Vukas D, Ledic D, Grahovac G, Kolic Z, Rotim K, Vilendecic M. Clinical outcomes in patients after lumbar disk surgery with annular reinforcement device: two-year follow up. Acta Clin Croat. 2013;52(1):87-91.
- Thome C, Klassen PD, Bouma GJ, et al. Annular closure in lumbar microdiscectomy for prevention of reherniation: a randomized clinical trial. Spine J. 2018;18(12):2278-2287.
- Cho PG, Shin DA, Park SH, Ji GY. Efficacy of a Novel Annular Closure Device after Lumbar Discectomy in Korean Patients : A 24-Month Follow-Up of a Randomized Controlled Trial. Journal of Korean Neurosurgical Society. 2019;62(6):691-699.
- Lequin MB, Barth M, Thome C, Bouma GJ. Primary limited lumbar discectomy with an annulus closure device: one-year clinical and radiographic results from a prospective, multi-center study. Korean J Spine. 2012;9(4):340-347.
- Kursumovic A, Rath SA. Effectiveness of an annular closure device in a “real-world” population: stratification of registry data using screening criteria from a randomized controlled trial. Med Devices (Auckl). 2018;11:193-200.
- Kursumovic A, Rath S. Performance of an Annular Closure Device in a ‘Real-World’, Heterogeneous, At-Risk, Lumbar Discectomy Population. Cureus. 2017;9(11):e1824.
- Sanginov AJ, Krutko AV, Baykov ES, Lutsik AA. Outcomes of surgical treatment of lumbar disc herniation using an annular closure device. Coluna/Columna. 2018;17(3):188-194.
- Ardeshiri A, Miller LE, Thome C. Two-year real-world results of lumbar discectomy with bone-anchored annular closure in patients at high risk of reherniation. Eur Spine J. 2019.
- Klassen PD, Bernstein DT, Kohler HP, et al. Bone-anchored annular closure following lumbar discectomy reduces risk of complications and reoperations within 90 days of discharge. J Pain Res. 2017;10:2047-2055.
- Kienzler JC, Klassen PD, Miller LE, et al. Three-year results from a randomized trial of lumbar discectomy with annulus fibrosus occlusion in patients at high risk for reherniation. Acta Neurochir (Wien). 2019.
- Nanda D, Arts MP, Miller LE, et al. Annular closure device lowers reoperation risk 4 years after lumbar discectomy. Med Devices (Auckl). 2019;12:327-335.
- FDA Summary of Safety and Effectiveness Data (SSED). Barricaid® Anular Closure Device (ACD). PMA P160050. https://www.accessdata.fda.gov/cdrh_docs/pdf16/P160050B.pdf.
- Malter AD, Larson EB, Urban N, Deyo RA. Cost-effectiveness of lumbar discectomy for the treatment of herniated intervertebral disc. Spine (Phila Pa 1976). 1996;21(9):1048-1054; discussion 1055.
- Tosteson AN, Skinner JS, Tosteson TD, et al. The cost effectiveness of surgical versus nonoperative treatment for lumbar disc herniation over two years: evidence from the Spine Patient Outcomes Research Trial (SPORT). Spine (Phila Pa 1976). 2008;33(19):2108-2115.
- Tosteson AN, Tosteson TD, Lurie JD, et al. Comparative effectiveness evidence from the spine patient outcomes research trial: surgical versus nonoperative care for spinal stenosis, degenerative spondylolisthesis, and intervertebral disc herniation. Spine (Phila Pa 1976). 2011;36(24):2061-2068.
- Koenig L, Dall TM, Gu Q, Saavoss J, Schafer MF. How does accounting for worker productivity affect the measured cost-effectiveness of lumbar discectomy? Clin Orthop Relat Res. 2014;472(4):1069-1079.
- Heindel P, Tuchman A, Hsieh PC, et al. Reoperation Rates After Single-level Lumbar Discectomy. Spine (Phila Pa 1976). 2017;42(8):E496-E501.
- Gray DT, Deyo RA, Kreuter W, et al. Population-based trends in volumes and rates of ambulatory lumbar spine surgery. Spine (Phila Pa 1976). 2006;31(17):1957-1963; discussion 1964.
- Ament J, Thaci B, Yang Z, et al. Cost-effectiveness of a Bone-anchored Annular Closure Device Versus Conventional Lumbar Discectomy in Treating Lumbar Disc Herniations. Spine (Phila Pa 1976). 2019;44(1):5-16.
Disclosures and COI: Morgan Lorio, MD, FACS: None. Choll Kim: Consultant for Elliquence. Paid Member of Mock FDA Advisory Panel for Intrinsic Therapeutics. Ali Araghi, DO: Consulting fees from Intrinsic Therapeutics. Jason Inzana, PhD: salaried employee of Telos Partners, LLC, which received payments from ISASS to support literature review and manuscript development. Telos Partners, LLC has received fees unrelated to this manuscript from entities in the spine space, including Zimmer Biomet, Orthopediatrics, Intrinsic Therapeutics, K2M, Medacta International, Merit Medical, Wenzel Spine, and SiNtx Technologies. James Yue, MD: Consultant for Elliquence Spine, Vertical Spine, Aesculap Spine.
